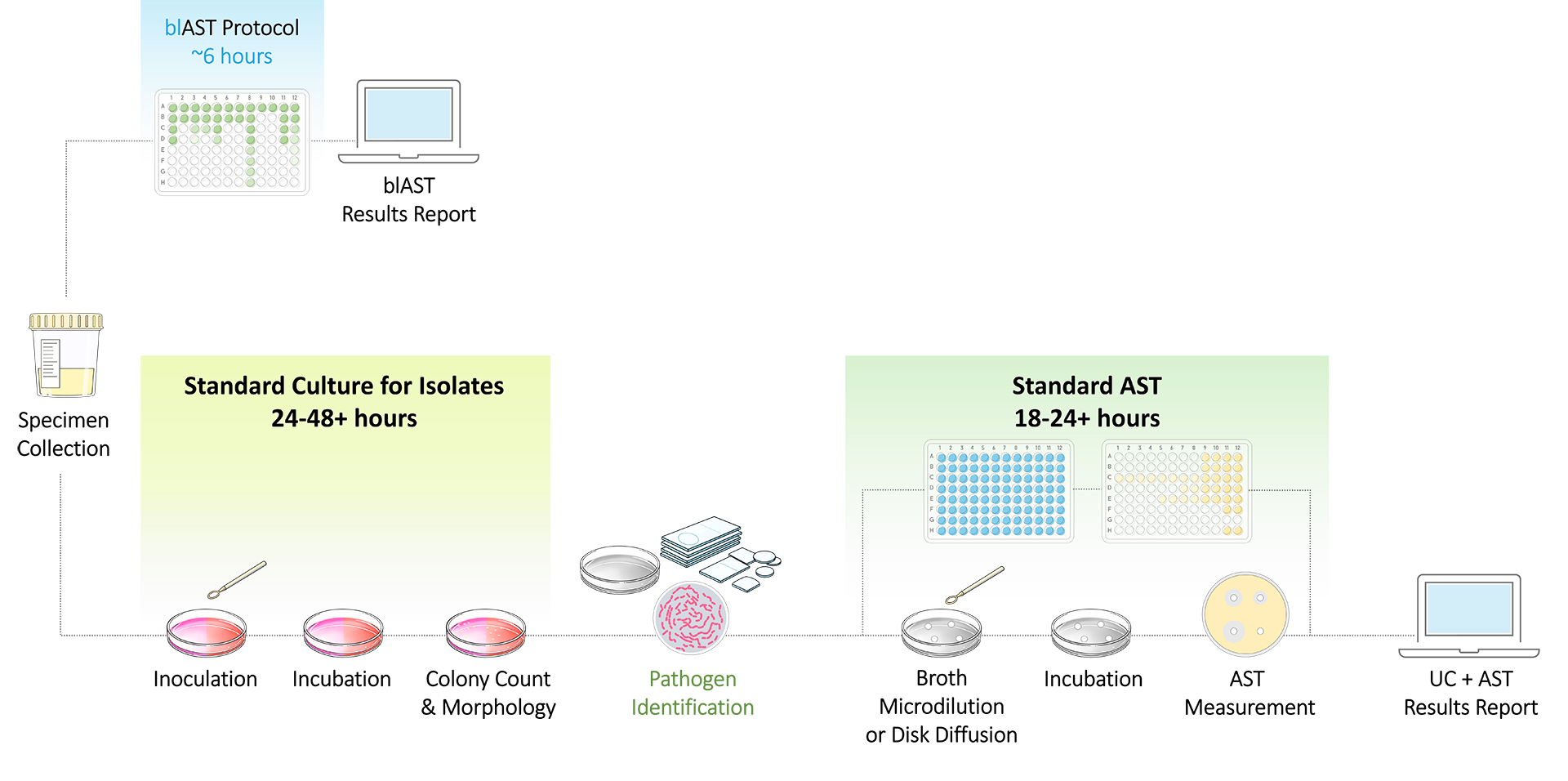
What is blAST?
blAST is a Bio-Labeled Antimicrobial Susceptibility Test (AST) that uses award-winning click chemistry to analyze antimicrobial sensitivity directly from urine samples.
Can AST be done in less
than 24 hours?
blAST is a direct urine phenotypic AST that provides single-shift results in ~6 hours.
blAST bypasses the need for a growth phase and eliminates standard culture timelines. The simplified laboratory workflow provides actionable, accurate, and reliable AST results from metabolically-active pathogens.